NOTICIAS
BOAS PRÁTICAS de FARMACOVIGILÂNCIA : ” Formação completa, contempla os pontos principais de Boas Práticas de Farmacovigilância “
Decorreu nos dias 24 e 25 de março a formação BOAS PRÁTICAS de FARMACOVIGILÂNCIA bajo o lema " Análise do conjunto de regulamentações, documentos e informações que permitem ao titular de AIM avaliar a sua área de farmacovigilância face as exigências regulamentares....
New EMA GMP Guidelines in the next three Years : The 3-year work plan for the Inspectors Working Group
The European Medicines Agency (EMA) has published a new version of the '3-year work plan for the Inspectors Working Group' covering the period from January 2026 to December 2028. This workplan sets out the routine activities of the GMDP IWG that contribute to the...
Formação QUALITY MANAGEMENT SYSTEM: ” Ótima forma de rever temas e aprofundar conhecimentos com casos do dia -a-dia “
“ Formação muito clara e prática acerca de vários temas da gestão da qualidade . “ GENERIS Nos dias 23 e 24 de março, decorreu em Lisboa a formação QUALITY MANAGEMENT SYSTEMS, bajo o lema " Uma visão completa e essencial na aprendizagem dos conceitos e...
C(2026) 1809 expands the list of Class IIb implantable devices exempt from the obligation to perform an assessment of technical documentation for every device under Article 52(4).
Well established technologies (WET) are relatively simple devices, having common and stable designs with little evolution, well-known safety and clinical performance characteristics and a long history on the market. Criteria for identifying WET have been set in...
EU PARLIAMENT Textes Adopted on AI – 26/03/2026 Simplification of the implementation of harmonised rules on artificial intelligence (Digital Omnibus on AI)
The document contains a comprehensive set of amendments aimed at simplifying the EU AI Act’s implementation, improving proportionality for SMEs/SMCs, reinforcing AI Office powers, prohibiting harmful AI misuse (like sexual deepfakes), clarifying conformity...
Registo Investigação e Tratamento de RECLAMAÇÕES : “Muito útil para melhorar os processos já implementados “
No passado 20 de março, decorreu em Lisboa uma nova edição da formação exlusiva da Formiventos sobre " Registo Investigação e Tratamento de RECLAMAÇÕES de QUALIDADE e de DISTRIBUIÇÃO", conduzida por Sónia Rei, Associate Director Operations, na Hikma A iniciativa...
ISO 20417:2026 – Updated Guidance on Medical Device Information – The second edition of ISO 20417:2026 (Medical devices – Information to be supplied by the manufacturer) has just been published.
This edition cancels and replaces the first edition (ISO 20417:2021), which has been technically revised. ISO 20417:2026 sets out requirements for the information that manufacturers must supply with medical devices and their accessories, including labelling,...
COMMISSION DELEGATED REGULATION (EU) …/… amending Regulation (EU) 2017/745 of the European Parliament and of the Council as regards the list of implantable devices and class III devices exempted from the obligation to perform clinical investigations C/2026/1798 final
Well established technologies (WET) are relatively simple devices, having common and stable designs with little evolution, well-known safety and clinical performance characteristics and a long history on the market. Criteria for identifying WET have been set in...
Progressing EMA’s PRIority Medicines scheme through new pilot features
Report on the experience with the new PRIME features following the 5-year review of the scheme to support the development of medicines targeting unmet needs This report presents the findings from a two-year pilot (April 2023–March 2025) to assess the...
FDA Releases Draft Guidance on Alternatives to Animal Testing in Drug Development
The U.S. Food and Drug Administration today issued a draft guidance intended to help drug developers validate new approach methodologies (NAMs) to be used instead of animal testing in drug development, and to bring safe, effective drugs to market sooner based on...
‘Pharma package’: Council and Parliament reach a deal on new rules for a fairer and more competitive EU pharmaceutical sector
The Council and the European Parliament have reached an agreement on the ‘pharma package’, a new set of rules that will increase patients' access to medicine and make the EU’s pharmaceutical sector fairer and more competitive. The package represents a far-reaching...
EU Pharma Package: Final Texts officially published
The Committee of Permanent Representatives of the EU Member States (COREPER I) has approved the trilogue outcome reached in December 2025. The final texts have now been officially published: The new DIRECTIVE OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL on the Union...
EU COMMISSION Artificial Intelligence Act – detailed arrangements on evaluations and proceedings.
The European Union continues to take important steps in shaping the future of responsible AI governance. The European Commission has launched a new initiative under the Artificial Intelligence Act to define the detailed arrangements for evaluations and proceedings...
GDP Nível 2 : “Muito útil para uma visão mais abrangente dos riscos e das oportunidades de melhoria e simplificação internas
Decorreu na semana passada mais uma edição da formação exclusiva de Formiventos :GDP Nível 2 : BOAS PRÁTICAS DE DISTRIBUIÇÃO Nível Avançado Uma oportunidade perfeita para consolidar e completar a sua formação anterior em Boas Práticas de Distribuição, que permite...
MEDICAL DEVICES : SIMPLIFYING THE RULES
On 16 December 2025, the European Commission put forward a proposal for a regulation to simplify the rules governing medical devices in the EU. The proposal forms part of a broader package of measures aimed at improving citizens' health and strengthening the...
EDPB and EDPS adopted Joint Opinion 3/2026 on the proposed European Biotech Act
On the Proposal for a Regulation on establishing a framework of measures for strengthening Union’s biotechnology and biomanufacturing sectors particularly in the area of health (European Biotech Act) Adopted on 10 March 2026
FDA Biosimilar Development – Q&A Guidance: Q&As on Biosimilar Development and the BPCI Act
The U.S. Food and Drug Administration has issued a guidance document titled “Q&As on Biosimilar Development and the BPCI Act.” This guidance provides FDA’s current thinking on regulatory and scientific considerations for developing biosimilar products under the...
NDSG workplan 2026-2028 : Data and AI in medicines regulation
The joint HMA/EMA Network Data Steering Group (NDSG) is the strategic advisory group established to maximise data interoperability and exchange, to improve access to data and evidence generation, and to leverage Artificial Intelligence (AI) for the benefit of public...
New guidance on the conduct of clinical trials during public health emergencies
Just published a draft guidance detailing how clinical trials should be designed and conducted when public health emergencies arise. The document is aimed at sponsors and all parties involved in clinical research across the EU. The document provides practical...
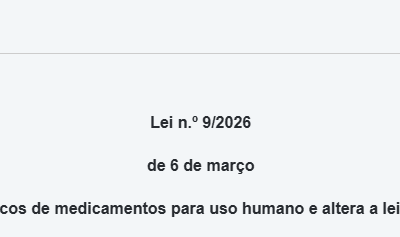
Nova lei regula ensaios clínicos de medicamentos para uso humano em Portugal
publicado no site infarmed Foi publicada em Diário da República a Lei n.º 9/2026, de 6 de março, diploma que assegura a execução, na ordem jurídica nacional, do Regulamento (UE) n.º 536/2014 relativo aos ensaios clínicos, e procede à terceira alteração à Lei da...
FDA draft guidance :“New Clinical Investigation Exclusivity: 3-Year Exclusivity for Drug Products – Questions and Answers.”
This guidance provides FDA’s current thinking on the scope and application of 3-year exclusivity under section 505(c)(3)(E)(iii) and 505(j)(5)(F)(iii) of the FD&C Act for drug products approved with new clinical investigations (other than bioavailability studies)...
webinar INTELIGÊNCIA ARTIFICIAL NA INDÚSTRIA FARMACÊUTICA :IA EM CONTEXTO GxP (Annex 11, 22, EU AI Act , GAMP®)
“Formação bastante esclarecedora, em que a informação foi apresentada de forma clara e bem resumida servindo de bom ponto de partida para aprofundar conhecimentos no papel da IA na indústria farmacêutica. Foram também apresentados exemplos práticos o que permitiu...
GESTÃO da DISPONIBILIDADE do MEDICAMENTO: “Formação muito útil !
A Formiventos apresentou no passado dia 27 uma nova edição da formação : GESTÃO da DISPONIBILIDADE do MEDICAMENTO ✅Foi uma formação com uma forte componente prática, com o objetivo de alargar a experiência e o conhecimento relativo aos pontos críticos das...
IMDRF Playbook for Medical Device Regulatory Reliance Programs
The information in this document applies to all medical devices, including IVD medical devices. Unless otherwise specified, the principles discussed can be applied to any phase of the product lifecycle (e.g., technical documentation2 review, evaluation of quality...
IMDRF Considerations for the selection of IMDRF Adverse Event Terminology
MDRF International Medical Device Regulators Forum. Considerations for the selection of IMDRF Adverse Event Terminology A Guide for Industry Partners and Healthcare Providers. 25 February 2026 Scope: This document is intended to provide guidance on the correct...
Concept paper on the revision of the guidelines on Good Manufacturing Practice for medicinal products – Annex 15 – Qualification and Validation
A concept paper on the revision of Annex 15 has been published on the EMA's website. One of the main objectives of the revision is to make the application of Annex 15 mandatory for active substances (APIs). Until now, its application has been optional for active...
FDA Draft Guidance General Considerations for Patient Preference Studies February 2026
The Food and Drug Administration (FDA, Agency, or we) is announcing the availability of a draft guidance for industry entitled “E22 General Considerations for Patient Preference Studies.” The draft guidance was prepared under the auspices of the International...
Open consultation: Medical devices regulations: targeted consultation on the indefinite recognition of CE marked devices
The Medicines and Healthcare products Regulatory Agency (MHRA) is inviting members of the public to provide their views on proposals for the approach to recognising CE marked medical devices in Great Britain. We welcome the views from anyone who will be impacted by...
Health Technology Assessment: HTA 2025 Annual Report
Health Technology Assessment: HTA Coordination Group publishes its 2025 Annual Report The Member State Coordination Group on Health Technology Assessment (HTACG) has published its 2025 Annual Report, providing an overview of the first year of application of the EU...
Guidance for applicants for the preparation of the ‘precise scope’ section of the variation application form
This guidance aims at supporting applicants in completing the 'Precise scope and Background for a change, and Justification for grouping, worksharing and classification for unforeseen changes (if applicable)' (hereinafter called the 'Precise scope') section of the...
New Eurachem Guide to Quality in Analytical Chemistry Released – 4th Edition (2026)
The 4th edition of the Guide to Quality in Analytical Chemistry, has been published by Eurachem on 6 February 2026. This widely used guide provides laboratories with practical best-practice guidance for analytical operations, covering both qualitative and quantitative...
Harmonised ICH M15 Guideline, General Principles for Model-informed Drug Development, Adopted
The ICH M15 Guideline, General Principles for Model-informed Drug Development, has been adopted (Step 4 of the ICH harmonisation process) and will now enter the implementation phase (Step 5). Prior to the development of the M15 Guideline, a lack of common standards...
Computer Software Assurance for Production and Quality Management System Software
FDA is issuing this guidance to provide recommendations on computer software assurance for computers and automated data processing systems used as part of medical device production or the quality management system. This guidance: · Describes “computer software...
FDA CDER – 2025 New Drug Therapy Approvals: key takeaways
The FDA’s Center for Drug Evaluation and Research (CDER) has released its 15th annual New Drug Therapy Approvals report, offering a useful snapshot of the pace and direction of drug development in 2025. In 2025, CDER approved 46 new drugs never before approved or...
The MDSAP Audit Approach (MDSAP AU P0002.010) was just revised
Medical device/medtech regulatory updates this week: The MDSAP Audit Approach (MDSAP AU P0002.010) A few suggested next steps if you're impacted here: 1/ Audit your audit program. Your internal audit procedures and checklists likely reference QSR language and clause...
FDA Clinical Decision Support Software
This document supersedes “Clinical Decision Support Software” issued on January 6, 2026. The Food and Drug Administration (FDA) has long regulated software that meets the definition of a device in section 201(h) of the Federal Food, Drug, and Cosmetic...
FDA GUIDANCE : Cybersecurity in Medical Devices: Quality Management System Considerations and Content of Premarket Submissions
The U.S. Food and Drug Administration (FDA) has published “Cybersecurity in Medical Devices: Quality Management System (QMS) Considerations and Content of Premarket Submissions” updated guidance issued 3 February 2026. This guidance replaces the 2025 edition and...
The FDA’s updated compliance program manual
The FDA's updated compliance program manual page: https://www.fda.gov/medical-devices/quality-and-compliance-medical-devices/center-devices-and-radiological-health-cdrh-compliance-programs
The FDA’s Quality Management System Regulation (QMSR) is now LIVE and fully effective
Feb 02, 2026 marks the end of the 2‑year transition and the beginning of a globally aligned quality era, harmonized with ISO 13485:2016 👉 As of TODAY, all FDA-regulated companies must comply. If you manufacture, design, or distribute FDA-regulated medical...
ICH M11 guideline, clinical study protocol template and technical specifications – Scientific guideline
EMA Releases ICH M11: A New Era for Clinical Study Protocols The EMA has published the ICH M11 guideline, introducing a harmonised, structured and digital-ready template for clinical study protocols — a major step forward for global clinical research. 🔹...
Guidelines on key considerations in issuing recommendations on the classification of active substances as regards their supply (prescription and non-prescription status)
These guidelines provide an overview of the key considerations of the Committee of Experts on the Classification of Medicines as Regards their Supply (CD-P-PH/PHO), co-ordinated by the European Directorate for the Quality of Medicines & HealthCare (EDQM), Council...
COMMISSION IMPLEMENTING DECISION (EU) 2026/193 of 28 January 2026
📅 Adopted: 28 January 2026 📰 Published in the OJEU: 30 January 2026 Amending Implementing Decision (EU) 2021/1182 as regards harmonised standards for neurosurgical implants, biological evaluation of medical devices, clinical investigation of medical...
AUDITORIA A DOSSIERS DE AIM: “Formação clara, prática e com partilha de experiências”
Tendo em conta a alta prevalência de “inconformidades nos dossiers de AIM” nos mais diferentes procedimentos quando são submetidos, que leva naturalmente a problemas nos tempos de aprovação, a Formiventos apresentou no passado dia 28 uma nova edição da formação...
HERBAL FOOD SUPPLEMENTS .Guide for healthcare professionals
This guide was drafted by a multidisciplinary working group under the overall co-ordination of the Committee of Experts on Quality and Safety Standards in Pharmaceutical Practices and Pharmaceutical Care (CD-P-PH/PC). It provides: ✅ Structured recommendations...
M4Q(R2) The Common Technical Document for the Registration of Pharmaceuticals for Human Use: Quality
ICH HARMONISED GUIDELINE: The Common Technical Document for the Registration of Pharmaceuticals for Human Use: Quality SCOPE AND ORGANISATION The M4Q(R2) guideline establishes the location and structure of quality information for registration applications of all...
EUDAMED Production Release Jan 28th 2026
Release notes: https://lnkd.in/dmRB2inn User Manuals. Legacy devices: https://lnkd.in/d5cjaMgz Regulation devices: https://lnkd.in/dGSwt9rp
Guiding principles of good AI practice in drug development. January 2026
EU & US Regulatory News (AI in Drug Development – Guiding Principles) The European Medicines Agency (EMA), in collaboration with the U.S. Food and Drug Administration (FDA, has published Guiding Principles of Good AI Practice in Drug Development (January 2026)....
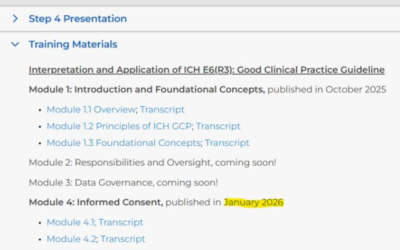
ICH E6(R3) update – New training materials available (Jan 2026)
The ICH Expert Working Group (EWG) has released additional training materials to support the implementation of ICH E6(R3), with a specific focus on Module 4: Informed Consent. Two transcripts are now available: 🔹 Module 4.1 (5 pages) Addresses key questions...
2025 EMA’s key recommendations in human medicines.
EMA have just published the overview of the 2025 EMA’s key recommendations in human medicines. It includes figures on the authorisation of medicines and a selection of new treatments that represent significant progress in their therapeutic areas. ✔️ 104...
Guiding principles of good AI practice in medicine development
EMA and the FDA have jointly identified 10 principles for good artificial intelligence practice in the medicines lifecycle. The use of AI technologies across the medicines lifecycle has increased significantly in recent years and holds great promise as a tool to...
European Pharmacopoeia publishes new data quality framework
The European Pharmacopoeia (Ph. Eur.) has published a new general chapter on Quality of data (5.38) in Issue 12.3, following its adoption by the European Pharmacopoeia Commission at its 182nd session in June 2025. New general chapter on quality of data 5.38 supports...