NOTICIAS
EMVO Systems Updates | May 2026
Explore the latest improvements across the European Medicines Verification System (EMVS), strengthening performance, usability, and patient safety across the network. If you want to learn more about this, please follow these links: https://lnkd.in/d_sJD77k...
EU agrees to simplify AI rules to boost innovation and ban ‘nudification’ apps to protect citizens
The European Commission welcomes the political agreement reached today between the European Parliament and the Council of the EU on simpler, innovation-friendly rules for artificial intelligence (AI). The Commission proposed the Digital Omnibus on AI only five months...
Formação DIGITAL PROMO REVIEW : ” Formação muito interativa , bem estruturada e com elevada partilha de casos práticos “
No passado 5 de maio , decorreu em Lisboa a sessão prática DIGITAL PROMO REVIEW: COMPLIANCE DIGITAL & SOCIAL MEDIA, conduzida por Sónia Ferreira, Technical Director & Local Scientific Manager na A Martins & Fernandes; bajo o lema " Uuma visão...
Best Practice Guidance for the Submission of Technical Documentation under Annex II and III of Medical Device Regulation
This best practice guidance document has been developed by members of Team NB who have reviewed the best practice guidance documents (exceeding twenty in number) submitted by individual Team NB notified body members, with the purpose to develop a unified approach on...
COMMISSION IMPLEMENTING REGULATION laying down certain uniform quality management and procedural requirements for the conformity assessment activities carried out by a notified body designated
Important development for the MDR and IVDR regulatory framework The Commission Implementing Regulation (EU) 2026/977 of 4 May 2026 - on Annex VII - has been published in the Official Journal of the European Union COMMISSION IMPLEMENTING REGULATION (EU)...
MASTER COURSE SUPLEMENTOS ALIMENTARES : “Evento muito bem organizado, conduzido e menistrado “
No passado 29 de abril de 2026 decorreu em Lisboa o Master Course Autorização, publicidade e comercialização de SUPLEMENTOS ALIMENTARES, bajo o lema "Uma visão exaustiva , atual e prática dos conhecimentos imprescindíveis para fabricar, introduzir e manter-se no...
Guidelines on the best practices for the traceability of medicines in hospital settings
The EDQM hospital medicine traceability guidelines aim at reducing medication errors and enhancing patient safety. It has been drafted by a Working Group and two Committees of Experts under the European Committee on Pharmaceuticals and Pharmaceutical Care (CD-P-PH),...
MDCG 2021-12 Rev.2 FAQ on the European Medical Device Nomenclature (EMDN) Revision 2 – April 2026
MDCG 2021-12 Rev.2: Updated FAQ on EMDN The revised MDCG 2021-12 Rev.2 FAQ on the European Medical Device Nomenclature (EMDN) is now available This document has been endorsed by the Medical Device Coordination Group (MDCG) established by Article 103 of Regulation (EU)...
GUIDELINE ON REQUIREMENTS FOR REVISION/RENEWAL OF CERTIFICATES OF SUITABILITY TO THE EUROPEAN PHARMACOPOEIA MONOGRAPHS
Certification of suitability to the Monographs of the European Pharmacopoeia GUIDELINE ON REQUIREMENTS FOR REVISION/RENEWAL OF CERTIFICATES OF SUITABILITY TO THE EUROPEAN PHARMACOPOEIA MONOGRAPHS The holder of a certificate of suitability (CEP) shall inform the...
EMA Clinical Data Publication (CDP)
Questions and Answers (Q&As) on the European Medicines Agency policy on the publication of clinical data for medicinal products for human use (Policy 0070) work-share initiative with Health Canada (HC) The work-share process is a voluntary collaborative...
PLANOS DE CONTINGÊNCIA em Farmacovigilância: “Excelente formação, muito prática”
No passado dia 21 de abril , teve lugar a sessão formativa PLANOS DE CONTINGÊNCIA /BUSINESS CONTINUITY em Farmacovigilância, conduzida por Ana Maria Tome, Senior Consultant GVP, na ADAMAS Consulting Nesta formação foram analisadas as chaves, os conteúdos e as...
Formação CTD MODULO 3 : ” Formação útil, clara e objetiva. Bons exemplos práticos para consolidar os conceitos abordados “
“Gostei muito da formação, achei muito enriquecedora e interessante . Gostei particularmente dos momentos de discussão e partilha de conhecimentos .” FRESENIUS-KABI Decorreu no passado dia 21 de abril, em Lisboa, a edição 2026 da formação CTD MODULO 3, conduzida...
📢 EU Releases Updated Borderline & Classification Manual (MDR/IVDR)
The European Commission has updated the manual on borderline and classification Regulation (EU) 2017/745 on medical devices (MDR) and Regulation (EU) 2017/746 on in vitro diagnostic medical devices (IVDR)(Version 5, April 2026) in April 2026 to include...
Novo regulamento para notificação de efeitos indesejáveis de produtos cosméticos
Foi publicada em Diário da República a Deliberação n.º 324/2026, de 18 de março, que aprova o regulamento relativo à comunicação de efeitos indesejáveis associados à utilização de produtos cosméticos. A deliberação, da responsabilidade do INFARMED vem reforçar os...
EUDAMED 2026 WORKSHOP : “Excelente formação, muito útil e prática “
Decorreu ontem, dia 21 de abril, o WORKSHOP PC-BASED : EUDAMED 2026 , bajo o lema " Formação técnica e prática detalhada, centrada na utilização real da EUDAMED, para garantir a conformidade operacional total da sua empresa e dos seus produtos antes da sua...
New report on the state of AI readiness in healthcare across the European Union
This report draws on the 2024–2025 survey on AI for health in the WHO European Region, offering a focused lens on the EU. All 53 Member States of the WHO European Region received the survey, and 50 chose to participate (a 94% response rate), including all 27 EU Member...
Validation checklist for initial MAA under Regulation (EU) 2019/6 – biologicals other than immunologicals
This validation checklist is used by the Agency to validate initial marketing authorisation applications for pharmaceuticals and applicants should use it as a means to review in advance of their submission that standard requirements are fulfilled.
MDCG Guidance Update : Classification of Medical Devices – Revision 1
The Medical Device Coordination Group (MDCG) has published MDCG 2021-24 Rev.1 (April 2026), updating its guidance on the classification of medical devices under MDR (EU) 2017/745. This revision provides further clarification and practical examples to support...
AI Regulation in Pharmacovigilance. REGULATORY BRIEFING 2026
Over the last year, something shifted. The EMA–FDA joint Good AI Practice Principles (Jan 2026), the CIOMS Working Group XIV final report (Dec 2025), and the EU AI Act high-risk rules taking effect this August have moved AI in PV from "interesting pilot" to "regulated...
ICH Quality Risk Management as part of Integrated Quality Management
This presentation relates to Annex II.1 of ICH Q9(R1) To ensure the adequate management of risks and continuous improvement, it is essential for the principles and practices of QRM to be fully integrated within the PQS as outlined by ICH Q9 (R1). • ICH Q9 (R1) is not...
FDA Examples of Real-World Evidence Used in Medical Device Regulatory Decisions
This report presents 73 examples of market authorizations from fiscal years (FY) 2020-2025 building on the foundational work presented in the 2021 publication “Examples of Real-World Evidence (RWE) Used in Medical Device Regulatory Decisions,” which documented 90...
FDA eMDR – Electronic Medical Device Reporting
Update: April 10, 2026 The FDA has updated its Electronic Medical Device Reporting (eMDR) framework, confirming the transition to the ESG NextGen Unified Submission Portal As of April 14, 2025, the ESG NextGen portal replaces the legacy WebTrader interface, providing...
ACT EU Draft Guidance on Clinical Trials During Public Health Emergencies
European Commission and HMA published the materials from the ACT EU webinar (8 April) presenting the new draft guidance on the conduct of clinical trials during public health emergencies (PHEs) — Now open for public consultation until 30 April 2026.
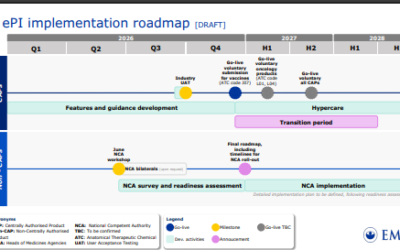
EMA’s ePI Roadmap
The European Medicines Agency (EMA) has released a draft roadmap for the implementation of electronic Product Information (ePI). This marks a major shift from traditional Word/PDF documents to a fully digitized, structured format designed to improve accessibility and...
Sponsor Frequently Asked Questions Clinical Trial Information System (CTIS) Frequently Asked Questions (FAQs) on the Sponsor’s workspace
This document is based on questions frequently raised to EMA by sponsors during CTIS events such as Walk-in Clinics, Bitesize Talks, and through the EMA CTIS Service Desk. It serves as complementary document to the Sponsor Handbook, which is the main operational...
The new CTIS Sponsor Handbook (v6.2, March 2026)
The EMA Clinical Trials Information System (CTIS) Sponsor Handbook provides clinical trial (CT) sponsors representing pharmaceutical industry, SME (small and medium-sized enterprises), academia, research organisations and other clinical trial sponsor organisations...
New revision of the Q&A document on art. 10(a) interruption of supply published
Q&A on practical aspects related to the implementation of the Article 10a obligation in case of interruption or discontinuation of supply of certain devices as introduced by Regulation (EU) 2024/1860 amending Regulations (EU) 2017/745 and (EU) 2017/746 as regards...
IMDRF Technical Framework for Artificial Intelligence Life Cycle Management” (IMDRF AIML WG N93 – April 2026)
This document is intended for manufacturers that are developing AI-enabled medical devices to identify considerations as they make critical technical and governance choices. It is intended to provide considerations specific to where AI-enabled medical devices may...
New Harmonised Standards under MDR – April 2026
The latest publication of harmonised standards under the Medical Devices Regulation (MDR) – April 2026 is now available. These updates are essential for manufacturers and stakeholders to demonstrate compliance with the MDR, providing presumption of conformity with the...
EMA/CHMP: Updated Guideline on Active Substances published
In March 2026, the now final "Guideline on the chemistry of active substances" was published on the EMA website. As reported in the news "EMA/CHMP: New Guideline Draft on Active Substances published for Comment", the draft guideline was open for public consultation...
Data Quality Framework for EU medicines regulation: application to Real-World Data
EMA released the "Data Quality Framework for EU medicines regulation: application to Real-World Data", establishing the principles and definitions under the scope of datasets used in medicine regulatory use cases. This document describes the Real-World Data...
BOAS PRÁTICAS de FARMACOVIGILÂNCIA : ” Formação completa, contempla os pontos principais de Boas Práticas de Farmacovigilância “
Decorreu nos dias 24 e 25 de março a formação BOAS PRÁTICAS de FARMACOVIGILÂNCIA bajo o lema " Análise do conjunto de regulamentações, documentos e informações que permitem ao titular de AIM avaliar a sua área de farmacovigilância face as exigências regulamentares....
New EMA GMP Guidelines in the next three Years : The 3-year work plan for the Inspectors Working Group
The European Medicines Agency (EMA) has published a new version of the '3-year work plan for the Inspectors Working Group' covering the period from January 2026 to December 2028. This workplan sets out the routine activities of the GMDP IWG that contribute to the...
Formação QUALITY MANAGEMENT SYSTEM: ” Ótima forma de rever temas e aprofundar conhecimentos com casos do dia -a-dia “
“ Formação muito clara e prática acerca de vários temas da gestão da qualidade . “ GENERIS Nos dias 23 e 24 de março, decorreu em Lisboa a formação QUALITY MANAGEMENT SYSTEMS, bajo o lema " Uma visão completa e essencial na aprendizagem dos conceitos e...
C(2026) 1809 expands the list of Class IIb implantable devices exempt from the obligation to perform an assessment of technical documentation for every device under Article 52(4).
Well established technologies (WET) are relatively simple devices, having common and stable designs with little evolution, well-known safety and clinical performance characteristics and a long history on the market. Criteria for identifying WET have been set in...
EU PARLIAMENT Textes Adopted on AI – 26/03/2026 Simplification of the implementation of harmonised rules on artificial intelligence (Digital Omnibus on AI)
The document contains a comprehensive set of amendments aimed at simplifying the EU AI Act’s implementation, improving proportionality for SMEs/SMCs, reinforcing AI Office powers, prohibiting harmful AI misuse (like sexual deepfakes), clarifying conformity...
Registo Investigação e Tratamento de RECLAMAÇÕES : “Muito útil para melhorar os processos já implementados “
No passado 20 de março, decorreu em Lisboa uma nova edição da formação exlusiva da Formiventos sobre " Registo Investigação e Tratamento de RECLAMAÇÕES de QUALIDADE e de DISTRIBUIÇÃO", conduzida por Sónia Rei, Associate Director Operations, na Hikma A iniciativa...
ISO 20417:2026 – Updated Guidance on Medical Device Information – The second edition of ISO 20417:2026 (Medical devices – Information to be supplied by the manufacturer) has just been published.
This edition cancels and replaces the first edition (ISO 20417:2021), which has been technically revised. ISO 20417:2026 sets out requirements for the information that manufacturers must supply with medical devices and their accessories, including labelling,...
COMMISSION DELEGATED REGULATION (EU) …/… amending Regulation (EU) 2017/745 of the European Parliament and of the Council as regards the list of implantable devices and class III devices exempted from the obligation to perform clinical investigations C/2026/1798 final
Well established technologies (WET) are relatively simple devices, having common and stable designs with little evolution, well-known safety and clinical performance characteristics and a long history on the market. Criteria for identifying WET have been set in...
Progressing EMA’s PRIority Medicines scheme through new pilot features
Report on the experience with the new PRIME features following the 5-year review of the scheme to support the development of medicines targeting unmet needs This report presents the findings from a two-year pilot (April 2023–March 2025) to assess the...
FDA Releases Draft Guidance on Alternatives to Animal Testing in Drug Development
The U.S. Food and Drug Administration today issued a draft guidance intended to help drug developers validate new approach methodologies (NAMs) to be used instead of animal testing in drug development, and to bring safe, effective drugs to market sooner based on...
‘Pharma package’: Council and Parliament reach a deal on new rules for a fairer and more competitive EU pharmaceutical sector
The Council and the European Parliament have reached an agreement on the ‘pharma package’, a new set of rules that will increase patients' access to medicine and make the EU’s pharmaceutical sector fairer and more competitive. The package represents a far-reaching...
EU Pharma Package: Final Texts officially published
The Committee of Permanent Representatives of the EU Member States (COREPER I) has approved the trilogue outcome reached in December 2025. The final texts have now been officially published: The new DIRECTIVE OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL on the Union...
EU COMMISSION Artificial Intelligence Act – detailed arrangements on evaluations and proceedings.
The European Union continues to take important steps in shaping the future of responsible AI governance. The European Commission has launched a new initiative under the Artificial Intelligence Act to define the detailed arrangements for evaluations and proceedings...
GDP Nível 2 : “Muito útil para uma visão mais abrangente dos riscos e das oportunidades de melhoria e simplificação internas
Decorreu na semana passada mais uma edição da formação exclusiva de Formiventos :GDP Nível 2 : BOAS PRÁTICAS DE DISTRIBUIÇÃO Nível Avançado Uma oportunidade perfeita para consolidar e completar a sua formação anterior em Boas Práticas de Distribuição, que permite...
MEDICAL DEVICES : SIMPLIFYING THE RULES
On 16 December 2025, the European Commission put forward a proposal for a regulation to simplify the rules governing medical devices in the EU. The proposal forms part of a broader package of measures aimed at improving citizens' health and strengthening the...
EDPB and EDPS adopted Joint Opinion 3/2026 on the proposed European Biotech Act
On the Proposal for a Regulation on establishing a framework of measures for strengthening Union’s biotechnology and biomanufacturing sectors particularly in the area of health (European Biotech Act) Adopted on 10 March 2026
FDA Biosimilar Development – Q&A Guidance: Q&As on Biosimilar Development and the BPCI Act
The U.S. Food and Drug Administration has issued a guidance document titled “Q&As on Biosimilar Development and the BPCI Act.” This guidance provides FDA’s current thinking on regulatory and scientific considerations for developing biosimilar products under the...
NDSG workplan 2026-2028 : Data and AI in medicines regulation
The joint HMA/EMA Network Data Steering Group (NDSG) is the strategic advisory group established to maximise data interoperability and exchange, to improve access to data and evidence generation, and to leverage Artificial Intelligence (AI) for the benefit of public...
New guidance on the conduct of clinical trials during public health emergencies
Just published a draft guidance detailing how clinical trials should be designed and conducted when public health emergencies arise. The document is aimed at sponsors and all parties involved in clinical research across the EU. The document provides practical...
Nova lei regula ensaios clínicos de medicamentos para uso humano em Portugal
publicado no site infarmed Foi publicada em Diário da República a Lei n.º 9/2026, de 6 de março, diploma que assegura a execução, na ordem jurídica nacional, do Regulamento (UE) n.º 536/2014 relativo aos ensaios clínicos, e procede à terceira alteração à Lei da...